The global incidence of cancer continues to rise, with an estimated 19.2 million new cancer cases and approximately 10 million deaths in 2020 [1]. Although existing treatments such as chemotherapy, radiotherapy, and targeted therapy have achieved some progress, drug resistance and low survival rates in advanced cancer remain major challenges. In recent years, immunotherapy, particularly bispecific antibodies (bsAbs), has attracted considerable attention as an emerging cancer treatment strategy [2]. Bispecific antibodies can simultaneously target two different antigens or epitopes, enhancing the immune system’s recognition and killing of tumor cells. They demonstrate stronger targeting capabilities and greater clinical benefits than monoclonal antibodies, especially in patients with relapsed or refractory cancer [3].
In March of this year, Nature Reviews Drug Discovery (IF: 57.00) published a review titled “The present and future of bispecific antibodies for cancer therapy” [4].

The review describes the various roles and mechanisms of bsAbs in cell signaling, providing diverse strategies to enhance the immune system’s attack on tumors and offering new hope for cancer treatment. It also presents perspectives on next‑generation bispecific antibodies in early development, including trispecific antibodies, bispecific antibody prodrugs, bsAbs that induce tumor‑target degradation, and bsAbs acting as cytokine analogs.
Current Status of bsAbs for Cancer Therapy
Currently, more than 200 bispecific antibodies are in clinical development, with about 50% having entered Phase II and Phase III trials, and some bsAbs have already been approved [5]. Approximately three‑quarters of bsAbs are being developed for solid tumors, primarily through T‑cell engagers (TCEs, accounting for 33%) and bsAbs that function via immune checkpoint inhibition and/or immunomodulatory mechanisms.
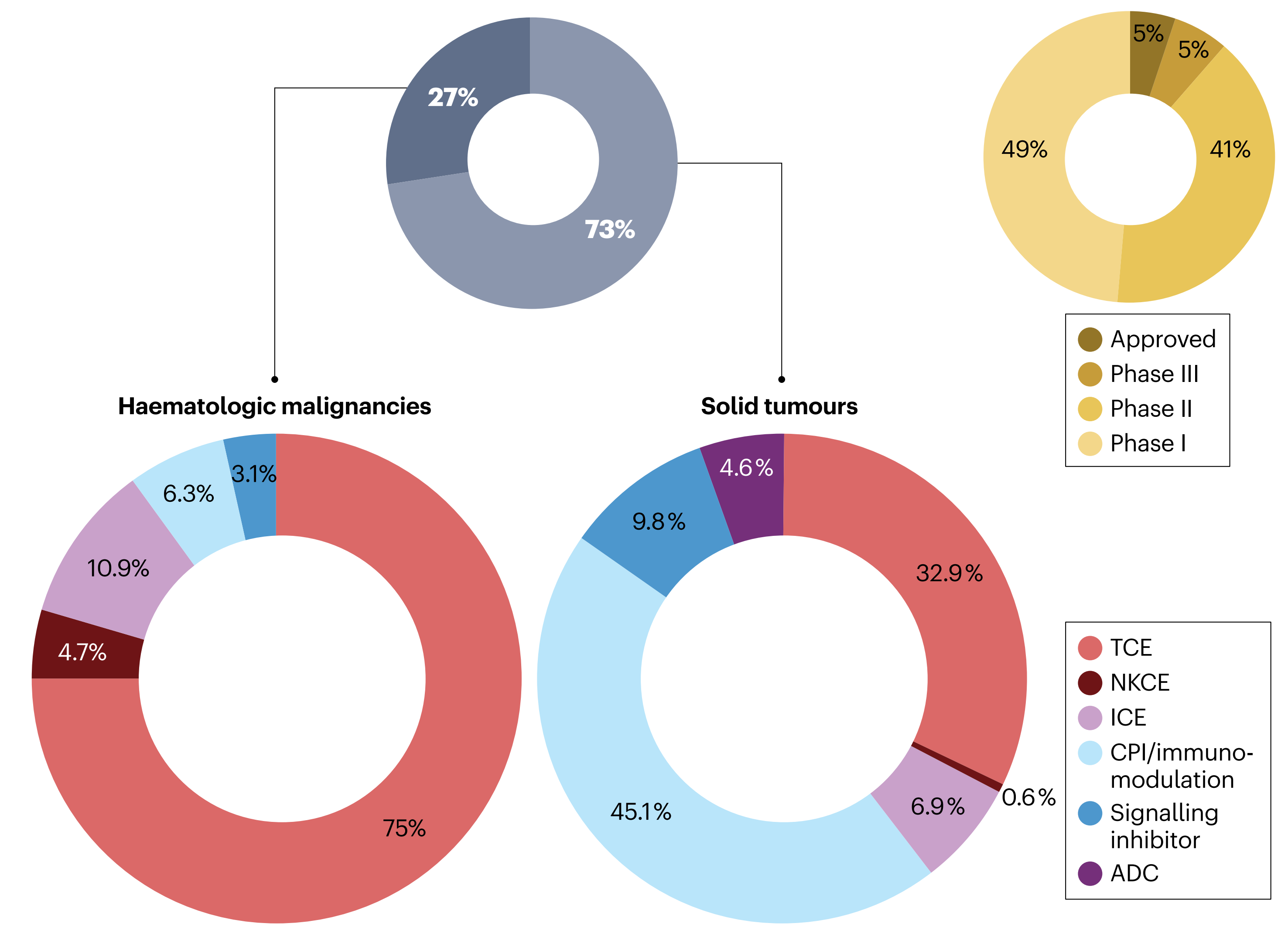
For hematologic malignancies, bsAbs mainly rely on TCEs and other immunomodulatory mechanisms. In addition, about 10% of bsAbs for solid tumors use dual signal inhibition, and bsAb antibody‑drug conjugates (ADCs) are also under development for solid tumors [6].
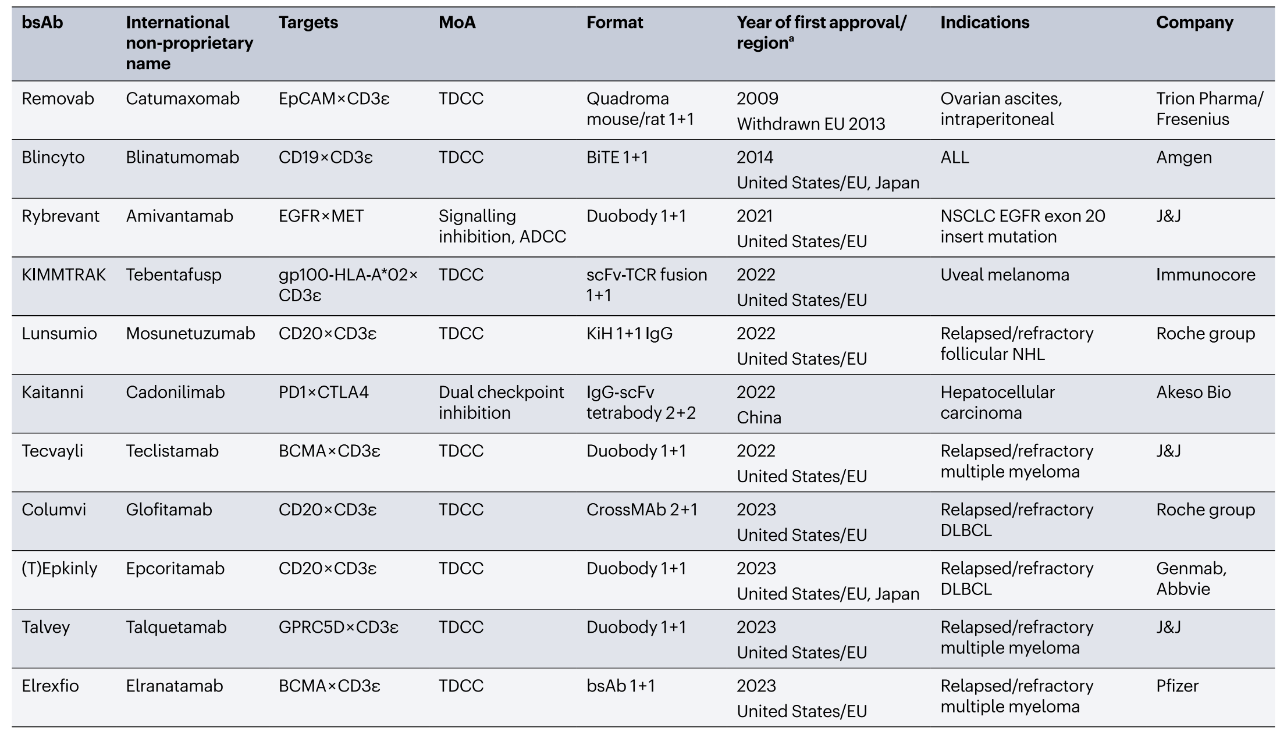
Table 1. Bispecific antibody drugs approved for cancer therapy
(Image placeholder – see original)
Mechanisms of Action of bsAbs
01 Dual Receptor Inhibition
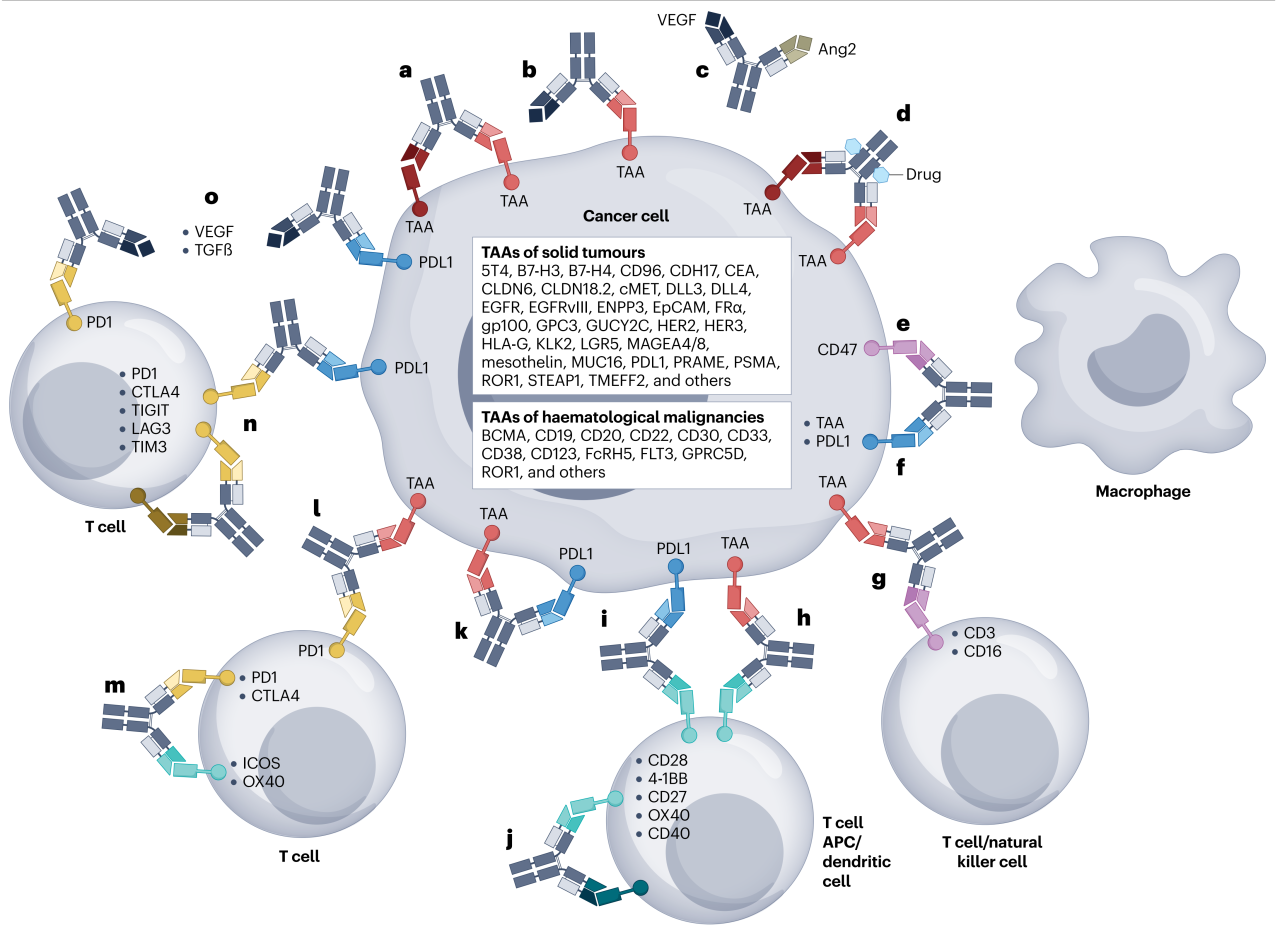
Bispecific antibodies provide an innovative therapeutic strategy by simultaneously modulating multiple cancer‑related signaling pathways, particularly those involving receptor tyrosine kinases (RTKs) and their associated receptors. Amivantamab (JNJ‑61186372), a bsAb targeting EGFR and MET, has been approved for the treatment of non‑small cell lung cancer (NSCLC) harboring EGFR exon 20 insertion mutations and has shown superior efficacy to chemotherapy alone [7].
In addition, bsAbs such as Petosemtamab (MCLA‑158) targeting EGFR and LGR5, and Zenocutuzumab (MCLA‑128) targeting HER2 and HER3, are in clinical development. Their development, combined with tumor organoid technology, is being evaluated in clinical trials for safety and efficacy in solid tumors [8].
02 Inhibition of Ligand‑Receptor Interactions
In cancer therapy, bsAbs can simultaneously block two different ligands or receptors, which is particularly effective for pathways involved in angiogenesis, such as vascular endothelial growth factor (VEGF) and angiopoietin 2 (ANG2). BI836880 is a trispecific nanobody (Nb) fusion protein composed of three nanobodies targeting VEGFA, ANG2, and HSA. This compound is currently in Phase I studies for patients with head and neck squamous cell carcinoma, NSCLC, and other solid tumors [9].
Several other bsAbs, such as Navicixizumab and Dilpacimab, have been designed to simultaneously target VEGF and DLL4, a cell surface ligand that activates the Notch‑1 receptor pathway and plays a central role in tumor angiogenesis. Clinical trials have shown that Navicixizumab in combination with chemotherapy exhibits good clinical efficacy with manageable toxicity [10].
03 Receptor Activation
In cancer immunotherapy, there are two main approaches to receptor activation: one is to fuse a receptor‑activating ligand (e.g., growth factor) to an antibody to form a fusion protein that specifically activates the receptor, triggering an immune response or cell death; the other is to use agonist antibodies to activate receptors, particularly death receptors of the tumor necrosis factor (TNF) superfamily or costimulatory receptors on immune cells [11].
However, monoclonal antibodies have yielded suboptimal results in clinical trials because ligands or receptors with costimulatory activity on immune cells often require cross‑linking of more than two receptors for activation. In contrast, bsAbs can simultaneously bind tumor‑associated antigens and TNF family receptors, effectively activating the receptor while avoiding interference from decoy receptors. For example, RG7386 is a tetravalent bsAb targeting DR5 and FAPα. It has shown potent antitumor activity in preclinical studies, and this approach can be extended to other costimulatory receptors for more effective immunotherapy [12].
04 bsAbs for Targeted Payload Delivery
Bispecific antibodies can deliver payloads (such as cytotoxic agents or radioactive substances) via two different methods. One is pre‑targeting therapy, where the bsAb first binds to tumor cells and then captures and delivers the radioactive or other payload. A new concept in this area is the SADA technology, which uses a modified p53 tetramerization domain to prolong payload retention in tumors [13]. The second method uses bispecific antibody‑drug conjugates (bsADCs) to deliver cytotoxic payloads to two epitopes or targets on the surface of tumor cells, enhancing the specificity of targeted therapy. Although both methods have their characteristics, the complexity of radio‑pre‑targeting limits its clinical application.
05 bsAbs for Cancer Immunotherapy
Most bsAbs for cancer immunotherapy either act through T‑cell‑driven natural or endogenous immunity (e.g., enhancing existing antitumor responses and/or overcoming checkpoint inhibition) or provide synthetic immunity through bsAb‑driven immune cell engagement, activation, and recruitment.
-
Checkpoint Inhibitors (CPIs)
These bsAbs enhance T‑cell‑mediated antitumor responses by simultaneously targeting immune checkpoint molecules (e.g., CTLA‑4, PD‑1/PD‑L1) and tumor‑associated antigens. Compared to monospecific antibodies, bsAbs can improve treatment specificity and efficacy while reducing side effects [14].
-
Effector Cell Engagers (TCEs and ICEs)
T cell engagers (TCEs) and innate immune cell engagers (ICEs) are bispecific antibodies that recruit T cells or other immune effector cells to the vicinity of tumor cells, using these immune cells to kill the tumor cells.
TCEs typically bind to both a tumor surface antigen and the T‑cell receptor (CD3ε chain), thereby directing T cells to kill tumor cells. Approved or clinically developed TCEs include blinatumomab (CD19 × CD3ε bsAb) for B‑cell precursor acute lymphoblastic leukemia [15]; mosunetuzumab (CD20 × CD3ε bsAb) for relapsed/refractory follicular non‑Hodgkin lymphoma; and teclistamab (BCMA × CD3ε bsAb) for multiple myeloma [16,17].
ICEs are another class of bispecific antibodies that bind to tumor surface antigens and activate natural killer (NK) cells to induce immune clearance of tumor cells. The most advanced NK cell engager is a short‑half‑life CD30 × CD16 bsAb for Hodgkin lymphoma, which binds to CD30‑positive Hodgkin lymphoma cells and activates NK cells, leading to tumor cell lysis [18].
-
Costimulatory bsAbs
Tumor‑targeting costimulatory bsAbs simultaneously bind tumor‑associated antigens (TAAs) and costimulatory receptors on immune cells (e.g., 4‑1BB, CD40, CD28), providing the necessary signal 2 within the tumor microenvironment (TME) to activate T cells and enhance antitumor immune responses [19]. bsAbs that activate dendritic cells (DCs) by binding CD40 and TAAs induce DC maturation and T‑cell activation, promoting tumor‑specific immune responses [20]. Dual costimulatory bsAbs target two different costimulatory receptors simultaneously, enhancing immune cell recruitment and activation. Finally, bsAbs that combine checkpoint inhibition and costimulation further enhance T‑cell immune responses and antitumor activity by simultaneously blocking inhibitory signals and providing costimulatory signals [21].
Future Perspectives
The authors suggest that future growth in multi‑specific antibody therapies may occur in four areas:
-
bsAbs targeting tumor‑associated RTKs with well‑defined dependencies;
-
bsADCs for tumor‑selective targeting with increased specificity;
-
bispecific PROTACs for targeted degradation of cell surface proteins;
-
multi‑specific antibodies for cancer immunotherapy.
In summary, significant progress has been made in the field of bispecific antibodies, with more than ten bsAbs approved and numerous clinical trials ongoing. Given the heterogeneity and adaptability of cancer, the current consensus is that no single approach is likely to become a universally applicable cancer immunotherapy. Therefore, future antibody therapies will need to be tailored to specific applications and combined with other therapies to maximize efficacy and safety.
Canton Biologics’ Bispecific Antibody Capabilities
Bispecific Antibody Therapy
Canton Biologics’ bispecific antibody platform has extensive experience in developing both symmetric and asymmetric configurations. It can complete development from DNA to IND within 12 months, with outstanding performance in expression level, yield, and purity. Average clone expression reaches approximately 10 g/L, with purity up to 99%.
In addition, the platform uses advanced 2D/3D sequence and structure analysis and computational tools for risk prediction and strategy optimization, ensuring a high success rate in antibody development. Canton Biologics provides reliable and efficient antibody solutions to its clients.
References (scroll to view)
[1] Lu L, S.Mullins C, Schafmayer C, et al. Global assessment of recent trends in gastrointestinal cancer and lifestyle risk factors. Cancer, 2022, 41(07):308-323.
[2] Brinkmann, U. & Kontermann, R. E. Bispecific antibodies. Science 372.
[3] Labrijn, A. F., Janmaat, M. L., Reichert, J. M. & Parren, P. Bispecific antibodies: 916–917 (2021).
[4] Klein C, Brinkmann U, Reichert M J, et al. The present and future of bispecific antibodies for cancer therapy. Nature Reviews Drug Discovery, 2024, 23(4):301-319.
[5] Wang, S. et al. The state of the art of bispecific antibodies for treating human malignancies. EMBO Mol. Med. 13, e14291 (2021).
[6] Antonarelli, G. et al. Research and clinical landscape of bispecific antibodies for the treatment of solid malignancies. Pharmaceuticals 14, 884 (2021).
[7] Neijssen, J. et al. Discovery of amivantamab (JNJ‑61186372), a bispecific antibody targeting EGFR and MET. J. Biol. Chem. 296, 100641 (2021).
[8] Herpers, B. et al. Functional patient‑derived organoid screenings identify MCLA‑158 as a therapeutic EGFR × LGR5 bispecific antibody with efficacy in epithelial tumors. Nat. Cancer 3, 418–436 (2022).
[9] Hofmann, I. et al. Pharmacodynamic and antitumor activity of BI 836880, a dual VEGF and angiopoietin 2 inhibitor, alone and combined with programmed cell death protein‑1 inhibition. J. Pharmacol. Exp. Ther. 384, 331–342 (2022).
[10] Li, J. L. & Harris, A. L. Crosstalk of VEGF and Notch pathways in tumour angiogenesis: therapeutic implications. Front. Biosci. 14, 3094–3110 (2009).
[11] Runbeck, E., Crescioli, S., Karagiannis, S. N. & Papa, S. Utilizing immunocytokines for cancer therapy. Antibodies 10, 10 (2021).
[12] Brunker, P. et al. RG7386, a novel tetravalent FAP‑DR5 antibody, effectively triggers FAP‑dependent, avidity‑driven DR5 hyperclustering and tumor cell apoptosis. Mol. Cancer Ther. 15, 946–957 (2016).
[13] Santich, B. H. et al. A self‑assembling and disassembling (SADA) bispecific antibody (bsAb) platform for curative two‑step pretargeted radioimmunotherapy. Clin. Cancer Res. 27, 532–541 (2021).
[14] Sharma, P. et al. Immune checkpoint therapy — current perspectives and future directions. Cell 186, 1652–1669 (2023).
[15] Bargou, R. et al. Tumor regression in cancer patients by very low doses of a T cell‑engaging antibody. Science 321, 974–977 (2008).
[16] Budde, L. E. et al. Safety and efficacy of mosunetuzumab, a bispecific antibody, in patients with relapsed or refractory follicular lymphoma: a single‑arm, multicentre, phase 2 study. Lancet Oncol. 23, 1055–1065 (2022).
[17] Hindie, E. Teclistamab in relapsed or refractory multiple myeloma. N. Engl. J. Med. 387, 1721 (2022).
[18] Reusch, U. et al. A novel tetravalent bispecific TandAb (CD30/CD16A) efficiently recruits NK cells for the lysis of CD30+ tumor cells. mAbs 6, 728–739 (2014).
[19] Claus, C., Ferrara‑Koller, C. & Klein, C. The emerging landscape of novel 4‑1BB (CD137) agonistic drugs for cancer immunotherapy. mAbs 15, 2167189 (2023).
[20] Muik, A. et al. DuoBody‑CD40 × 4‑1BB induces dendritic‑cell maturation and enhances T‑cell activation through conditional CD40 and 4‑1BB agonist activity. J. Immunother. Cancer 10, e004322 (2022).
[21] Qiao, Y. et al. Cancer immune therapy with PD‑1‑dependent CD137 co‑stimulation provides localized tumour killing without systemic toxicity. Nat. Commun. 12, 6360 (2021).

