The process of converting a DNA sequence into a correctly folded protein product is extremely complex, involving transcription, translation, post‑translational modifications, protein folding, and finally secretion. Problems at any of these steps can affect protein expression [1], leading to protein degradation, aggregation, and cytotoxicity [1].
Usually, the design and engineering of candidate therapeutic proteins, especially monoclonal and bispecific antibodies, aim primarily to increase affinity for the target protein or improve thermal stability, with little evaluation of their ease of expression in host cell lines such as CHO cells [2]. Low expression levels of biopharmaceutical candidate molecules in early development are one of the main reasons for early termination of product development.
Difficult‑to‑express therapeutic proteins are typically those where the rates of transcription, translation, and folding/assembly are mismatched, including toxins, enzymes, membrane proteins, some monoclonal and bispecific antibodies, and highly hydrophobic proteins [3,4]. Coordinating and optimizing the various interconnected dynamic processes – from gene sequence to secreted protein – is necessary to maximize product yield [3].
Characteristics of Difficult‑to‑Express Proteins
Difficult‑to‑express proteins (DTEs) generally have the following features:
-
Low expression levels, significantly below the typical g/L range, or even no expression [5];
-
Expression causes cell growth inhibition;
-
Poor clonal stability of engineered cells (e.g., CHO cells).
Canton Biologics’ Achievements
-
20+ projects delivered:
-
100% success rate in increasing expression levels;
-
Successfully increased recombinant protein expression from <0.5 g/L to >5 g/L.
-
-
High‑molecular‑weight protein series:
-
Multimeric proteins (~3000 kDa) expression >3 g/L;
-
Multivalent antibodies expression >10 g/L;
-
Helical extracellular matrix proteins expression >5 g/L.
-
Canton Biologics’ Solutions
To address this challenge, Canton Biologics has developed the DTEasy toolbox for difficult‑to‑express protein drugs. It integrates and optimizes each key stage of the DTE expression process, successfully increasing protein yield and helping protein drug molecules move smoothly toward industrialization.
01 Molecular Engineering Strategies
-
Introduce point mutations and optimize amino acid sequences based on amino acid sequence analysis and protein structure prediction.
-
Perform protein truncation or fuse with expression tags.
-
Screen suitable antibody VH/VL framework pairs and genetically engineer heavy and light chain sequences to improve the protein’s three‑dimensional structure and thermal stability.
02 Host Selection Strategies
Compared to other expression systems, mammalian cells have superior protein folding and disulfide bond formation capabilities [6]. Different mammalian cell lines also have different glycosylation capacities. For proteins with high requirements for glycosylation modifications, such as coagulation factors, Canton Biologics’ proprietary GlycoExpress® (GEX) cell line can be used.
Moreover, for certain tissue‑specific proteins, such as the extracellular domain of G‑protein coupled receptors (GPCRs), expression levels in GEX® cells can be up to ten times higher than in conventional CHO cell lines.
03 Vector Optimization Strategies
Using transcriptomic differential expression analysis, insert cis‑acting elements positively correlated with DTE yield into expression vectors, select multi‑copy vectors or vectors specifically designed for DTEs, or co‑express protein inhibitors, ligands, etc.
04 Process Development
During cell culture, proprietary additives can be added. Customized screening and culture processes for different cells can increase viable cell density and prolong culture time, ultimately increasing yield. DTEasy proprietary additives require only a single simple addition during culture and can increase expression by up to 3‑fold. Canton Biologics’ CBoost® enhanced process platform achieves more than double the protein expression in enhanced fed‑batch culture. In addition, because cells must balance protein expression and growth, exogenous protein expression places considerable stress on cells [7]. To address this, Canton Biologics uses gentler orbital‑shaking bioreactors, which are easy to operate, reduce mixing time, lower shear stress, and minimize foaming and contamination.
Case Study 1
Signal peptides are located at the N‑terminus of secreted proteins and direct newly synthesized proteins into the endoplasmic reticulum lumen. The signal peptide sequence is then cleaved by signal peptidase. Signal peptide screening is an important approach to enhance protein secretion. Different signal peptides can lead to different proportions of N‑terminal truncation of the fusion protein, which may affect the purity and activity of the fusion protein.
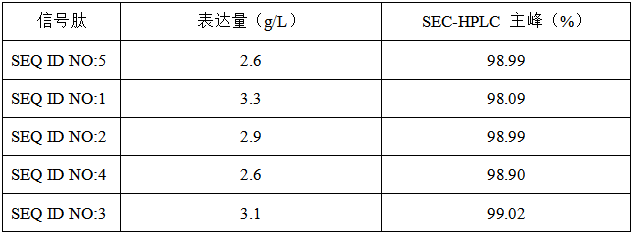
For a recombinant fusion protein Project A, Canton Biologics designed multiple signal peptides to enhance expression of the protein or fusion protein in host cells (results shown below; for details, see Canton Biologics patent: Use of signal peptide in expressing fusion protein, CN114539357B). Through combinations of different signal peptides, the purity and biological activity of the drug protein were significantly improved (Table 1).
Table 1. Expression level of recombinant fusion protein in cell culture supernatant and purity of fusion protein after purification
(Image placeholder – see original)
Case Study 2
For proteins that cause cytotoxicity when overexpressed, such as proteases and thrombin, co‑expression of their inhibitors can be considered to reduce the stress on cell survival and thereby increase protein yield.
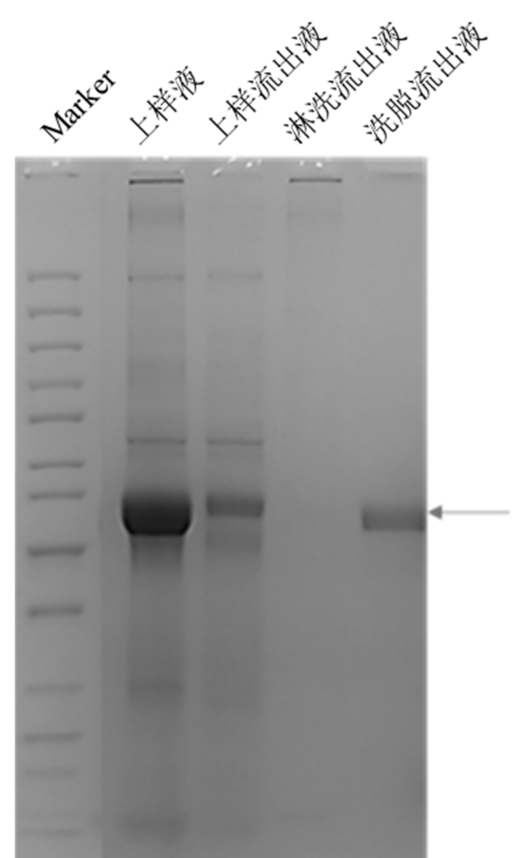
For tissue plasminogen activator (t‑PA), Canton Biologics constructed a dual‑expression vector co‑expressing t‑PA and a t‑PA inhibitor, as well as a one‑step chromatography purification process (results shown below; for details, see Canton Biologics patent: t‑PA purification method, CN114426962B). The non‑covalent complex formed between t‑PA and the t‑PA inhibitor has a loose structure and is easy to separate. Moreover, conventional t‑PA purification processes typically require 3–4 steps to achieve >90% purity. Co‑expression of the t‑PA inhibitor followed by lysine affinity chromatography enabled one‑step purification of t‑PA (Figure 1). The final purified t‑PA yield reached 361.5 mg/L, more than three times higher than existing technologies.
Figure 1. Lysine affinity chromatography results
(Image placeholder – see original)
Case Study 3
Commercial cell culture methods for producing sufficient proteins fall mainly into three categories: batch culture, fed‑batch culture, and continuous culture. In batch culture, all nutrients are provided in the initial medium; over time, nutrients decrease and metabolic inhibitors such as lactate increase. Fed‑batch culture involves intermittent or continuous addition of fresh medium during the culture process, avoiding inhibition of cells by metabolic products or nutrient depletion. Continuous culture continuously perfuses medium while removing waste, supplying nutrients, and harvesting product. Choosing the most appropriate culture method and supplementing with proprietary additives for different DTEs helps increase protein yield.
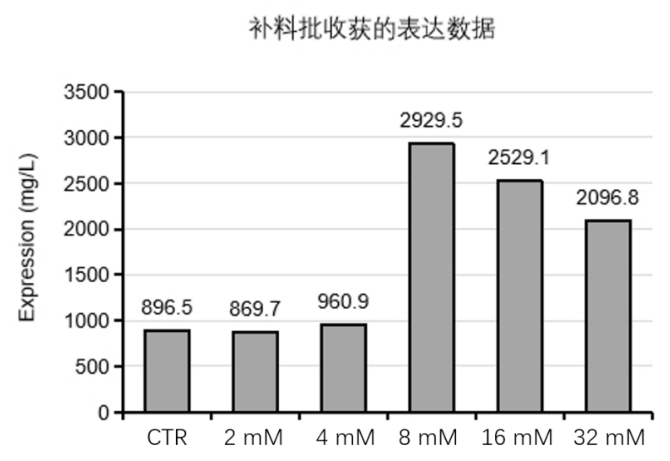
Previously, there was no efficient culture method for human nerve growth factor (hNGF), greatly limiting large‑scale production and clinical/pharmaceutical applications of hNGF. Canton Biologics used a fed‑batch culture method and discovered a new additive that increased the expression level of recombinant hNGF protein by at least 100%, with a maximum yield of approximately 2900 mg/L (Figure 2), greatly improving the expression level of hNGF in CHO cells (for details, see Canton Biologics patent: High‑efficiency expression method for recombinant nerve growth factor, CN2023108028220).
Figure 2. Bar chart of β‑NGF protein expression levels under media containing different concentrations of the additive
(Image placeholder – see original)
Case Study 4
Stirred‑tank bioreactors are commonly used in cell culture. However, the impeller can cause shear stress, and the submerged aeration method can generate foam, leading to even greater shear stress, which can damage cells or even cause cell death. In addition, scale‑up in stirred‑tank bioreactors involves many parameters and complex fluid dynamics, making scale‑up difficult or even unsuccessful.
Orbital‑shaking bioreactors mix cells through orbital shaking and can overcome the problems of stirred‑tank bioreactors. However, because the working principles are quite different, culture conditions for stirred‑tank bioreactors are not suitable for orbital‑shaking bioreactors. To address this, Canton Biologics successfully developed a cell culture method (for details, see Canton Biologics patent: Method for culturing cells using an orbital‑shaking bioreactor, CN113862224A). This method uses a gradient speed increase and temperature reduction strategy, along with appropriate medium optimization, resulting in higher cell viability and protein expression levels (Figures 3‑4).
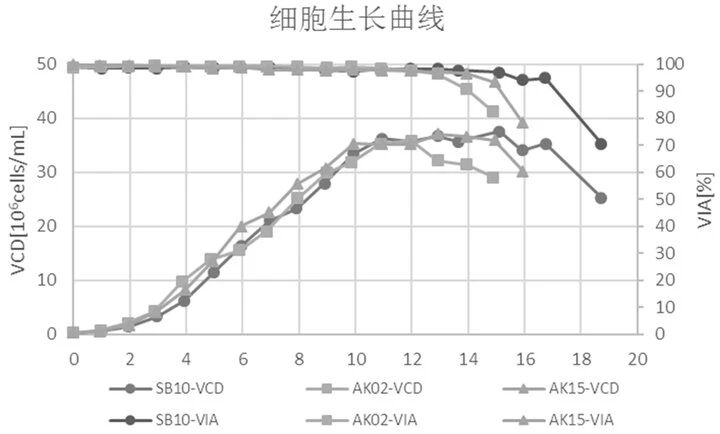
Figure 3. Viable cell density (VCD) and cell viability (VIA). SB‑10: orbital‑shaking bioreactor; AK02: 3 L stirred‑tank bioreactor; AK15: 15 L stirred‑tank bioreactor
(Image placeholder – see original)
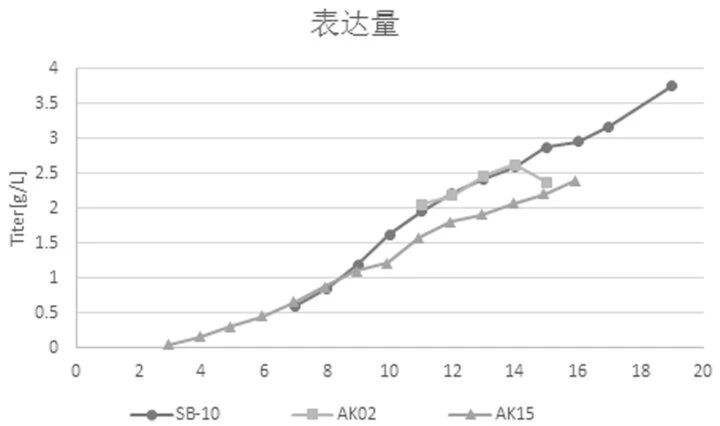
Figure 4. Protein expression levels. SB‑10: orbital‑shaking bioreactor; AK02: 3 L stirred‑tank bioreactor; AK15: 15 L stirred‑tank bioreactor
(Image placeholder – see original)
In summary, Canton Biologics’ DTEasy platform, through advanced molecular design, vector optimization, fine screening of host cell lines, and customized processes, has successfully solved the difficult‑to‑express problems of various protein types. We look forward to providing you with customized services!
Canton Biologics Patents (referenced in this article)
-
Use of signal peptide in expressing fusion protein [P]. Guangdong Province: CN202011358998.4, 2024-07-30.
-
t‑PA purification method [P]. Guangdong Province: CN202111574380.6, 2024-06-18.
-
High‑efficiency expression method for recombinant nerve growth factor [P]. Guangdong Province: CN202310802822.0, 2023-11-10.
-
Method for culturing cells using an orbital‑shaking bioreactor [P]. Guangdong Province: CN202010618579.3, 2021-12-31.
References (scroll to view)
-
Alves, C. S. & Dobrowsky, T. M. Strategies and Considerations for Improving Expression of “Difficult to Express” Proteins in CHO Cells. in Heterologous Protein Production in CHO Cells: Methods and Protocols (ed. Meleady, P.) 1–23 (Springer New York, New York, NY, 2017). doi:10.1007/978-1-4939-6972-2_1.
-
Szkodny, A. C. & Lee, K. H. A systemic approach to identifying sequence frameworks that decrease mAb production in a transient Chinese hamster ovary cell expression system. Biotechnol. Prog. e3466 (2024) doi:10.1002/btpr.3466.
-
Pybus, L. P. et al. Model‑directed engineering of ‘difficult‑to‑express’ monoclonal antibody production by Chinese hamster ovary cells. Biotechnol. Bioeng. 111, 372–385 (2014).
-
Chen, J.‑P. et al. Improving the soluble expression of difficult‑to‑express proteins in prokaryotic expression system via protein engineering and synthetic biology strategies. Metab. Eng. 78, 99–114 (2023).
-
L P, Greg D, R N W, et al. Model‑directed engineering of “difficult‑to‑express” monoclonal antibody production by Chinese hamster ovary cells. Biotechnol. Bioeng. 2014,111(2):372-85.
-
R.R. Burgess & Chen Wei. Protein Purification: Principles and Practice (2nd ed.). China Science and Technology Information, 1 (2013).
-
Nishimiya, D. Proteins improving recombinant antibody production in mammalian cells. Appl. Microbiol. Biotechnol. 98, 1031–1042 (2014).

