Respiratory syncytial virus (RSV) is a highly prevalent and contagious respiratory infection that can cause bronchiolitis and pneumonia, affecting more than 64 million people worldwide each year. Because the symptoms of RSV infection closely resemble those of influenza or other respiratory diseases, many infected individuals may not be correctly diagnosed. Young children, the elderly, and people with underlying health conditions are at high risk for severe RSV infection. Each year, RSV leads to more than 3 million hospitalizations of children under 5 years old globally, and nearly 60,000 children die from RSV infection. Based on current case data from Europe and the United States, the harm caused by RSV to young children even exceeds that of COVID‑19.

Recently, The Lancet Infectious Diseases (IF=36.4) published a review titled “Respiratory syncytial virus prevention within reach: the vaccine and monoclonal antibody landscape”, which outlines the clinical development of RSV vaccines and monoclonal antibodies, focusing on different target populations, antigens, and trial results [1].
The target populations for RSV prevention mainly fall into three categories: pediatric populations, pregnant women, and the elderly. For the pediatric population, the primary prevention strategies are passive immunoprophylaxis with monoclonal antibodies for infants under 6 months of age, and active immunization with live‑attenuated vaccines (LAVs) for older infants over 6 months of age. In addition, infants may be protected by passively transferred antibodies from vaccinated mothers. Currently, stabilized pre‑F subunit vaccines are in late‑stage development for administration to pregnant women. For the elderly population, three vaccination strategies (nucleic acid vaccines, subunit vaccines, and vector vaccines) are also in late‑stage clinical trials; all these vaccines use the pre‑F protein as the antigen [2].

Figure 1. RSV vaccines and monoclonal antibodies for infants, pregnant women, and the elderly
(Image placeholder – see original)
Currently, 33 RSV prevention candidates are in clinical development, involving six different approaches: recombinant vectors, subunits, particle‑based vaccines, live‑attenuated vaccines, chimeric vaccines, nucleic acid vaccines, and monoclonal antibodies. Among them, nine candidates are in Phase III clinical trials.
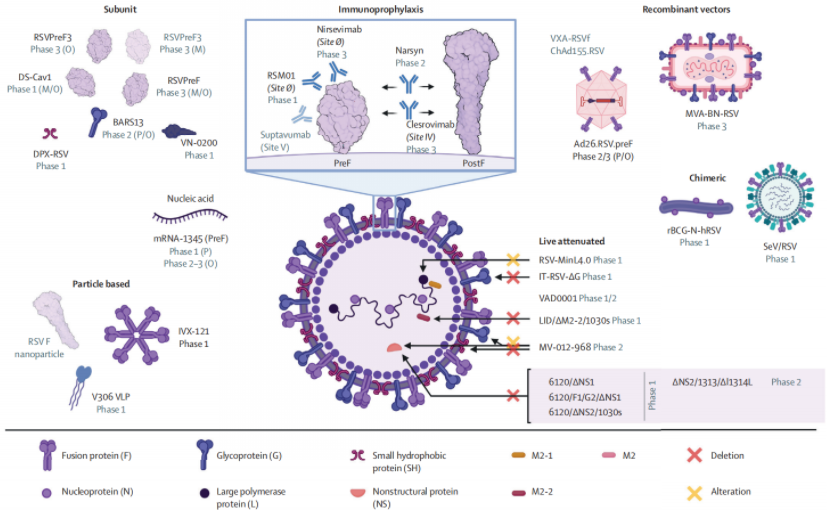
Figure 2. Overview of prophylactic vaccine candidates
(Image placeholder – see original)
01 Recombinant Vector Vaccines
Recombinant vector vaccines use modified replication‑defective viruses to deliver RSV antigen genes, inducing both humoral and cellular immunity. Currently, three candidates are in clinical development for pediatric and elderly populations.
MVA‑BN‑RSV and Ad26.RSV.pre‑F are two recombinant vector vaccines in development. Both have shown robust humoral and cellular immune responses, particularly in the elderly [3]. Phase II trials of MVA‑BN‑RSV demonstrated that the vaccine significantly reduces symptomatic RSV infection, with antibody responses lasting 6 months and safe boosting at 12 months.
Ad26.RSV.pre‑F showed good immunogenicity in Phase I and II trials, with 80% efficacy against RSV lower respiratory tract infection in the elderly, and was well‑tolerated in pediatric populations. Ad26.RSV.pre‑F has received FDA Breakthrough Therapy designation and has initiated Phase III trials to evaluate its efficacy in people aged 60 years and older. COVID‑19 adenovirus vector vaccines have revealed some potential safety concerns, such as immune thrombotic thrombocytopenia, which may affect the safety profile of RSV adenovirus vector vaccines.
02 Monoclonal Antibodies (mAbs)
Monoclonal antibodies are known as “magic bullets against infection” due to their high specificity for pathogens. Next‑generation RSV antibodies have been engineered with mutations in the Fc region that extend their half‑life, thereby providing all infants with protection against lower respiratory tract disease throughout the RSV season.
Nirsevimab is the leading monoclonal antibody candidate. Phase II and III trial results showed that nirsevimab is 70‑75% effective against RSV lower respiratory tract infection. Clesrovimab is an extended‑half‑life monoclonal antibody currently in Phase IIb/III and Phase III trials, and it has shown equivalent in vitro potency against RSV A and B strains [4].
For next‑generation monoclonal antibodies, key considerations include affordability and viral resistance monitoring. Costs can be reduced through high‑efficiency production, development of biosimilars, or local administration. However, the development of combination antibodies faces practical obstacles because each antibody must be registered separately. The epitopes targeted by monoclonal antibodies currently in development are highly conserved in nature, and antibody‑resistant strains have shown similar or lower viral fitness compared to non‑resistant strains in vitro.
03 Live‑Attenuated Vaccines (LAVs)
LAVs are designed to mimic natural infection, generating potent immune responses including local mucosal antibodies and cellular responses, while their pathogenicity is reduced through attenuation. A deep understanding of the RSV genome and its reverse genetics has enabled researchers to rationally design LAV candidates that limit viral replication by deleting or modifying key proteins that regulate RNA synthesis or interfere with host immune responses.
In a pooled analysis of seven Phase I trials (n=239, children aged 6‑24 months), LAV candidates were considered safe after the first dose, and no vaccine‑enhanced disease was observed. Estimated data showed that five LAV candidates were 67% effective against RSV acute respiratory disease and 88% effective against RSV lower respiratory tract infection. Immunological analysis indicated that a four‑fold increase in RSV plaque reduction neutralizing antibody titer was a good predictor of vaccine efficacy, and immune responses lasted for one year [5].
Overall, LAVs provide an important needle‑free, intranasal immunization tool for older infants, particularly those who cannot be fully protected by monoclonal antibodies or maternal vaccines. Further clinical development of LAVs is expected to directly improve child health by reducing pediatric infections and indirectly reduce infections in the elderly through herd immunity.
04 Chimeric Vaccines
Chimeric live virus vaccine candidates, which express RSV proteins in an attenuated virus, have shown good safety profiles. Compared to vector vaccine candidates, chimeric vaccines offer more favorable antigen presentation and effectively activate adaptive immune responses. Currently, two chimeric RSV vaccine candidates are in Phase I trials. The first uses a replication‑defective Sendai virus modified to express RSV F protein (SeV/RSV); the second uses a live‑attenuated recombinant bacillus Calmette‑Guérin (BCG) vector expressing RSV N protein (rBCG‑N‑hRSV), administered via the intradermal route.
05 Subunit Vaccines
Subunit vaccines are protein‑based vaccines. Due to the vaccine‑enhanced respiratory disease caused by the historical formalin‑inactivated RSV vaccine, this approach has been avoided in RSV‑naive children. However, five vaccine candidates using stabilized pre‑F protein as the antigen have made progress. Currently, eight subunit vaccine candidates are being developed for two distinct target populations: pregnant women and the elderly. The following discussion covers candidates using fusion antigens and non‑fusion antigens.
-
Pre‑F protein subunit vaccines
The DS‑Cav1 vaccine, developed by the US National Institutes of Health (NIH) and the National Institute of Allergy and Infectious Diseases (NIAID), demonstrated proof‑of‑concept for structure‑based vaccine design in a Phase I trial, with serum neutralizing activity increased more than 10‑fold after vaccination and sustained for one RSV season. In addition, RSVpre‑F is a bivalent (A and B subtypes) stabilized pre‑F protein vaccine without adjuvant. The Phase III MATISSE trial of RSVpre‑F is expected to be unblinded in 2023; related studies have shown 75% efficacy against RSV infection [6].
-
Non‑F protein antigen subunit vaccines
Three subunit vaccines based on non‑F protein antigens are currently in development. BARS13 uses the RSV G protein as an antigen and employs cyclosporine A to induce regulatory T cells; it was safe and immunogenic in Phase I trials and has now entered Phase II. DPX‑RSV uses the ectodomain of the RSV‑A small hydrophobic (SH) protein as an antigen and uses a lipid‑oil depot delivery platform to prolong exposure to the antigen and adjuvant; Phase I trials showed good safety and immunogenicity in adults aged 50‑64 years. VN‑0200 uses VAGA‑9001a as the antigen and MABH‑9002b as the adjuvant and is in Phase I trials [7].
06 Particle‑Based Vaccines
Particle‑based vaccines use particle assembly technology to display multiple antigens, offering significant immunological potential. IVX‑121 uses a self‑assembling synthetic virus‑like particle platform technology to display 20 trimeric stabilized pre‑F proteins (DsCav‑1). This nanoparticle is computationally designed to stabilize the pre‑F protein and modulate antigen density in vitro [8]. In preclinical studies, IVX‑121 elicited neutralizing antibody responses 10‑fold higher than DsCav1 alone. Phase I clinical trials were initiated in 2021, with first results expected in 2022. The company plans to develop a bivalent virus‑like particle vaccine containing antigens from both RSV and human metapneumovirus after completion of the monovalent RSV candidate trial. Overall, particle‑based vaccines are still in early development but have shown potential to elicit potent immune responses in pregnant women and the elderly.
07 Nucleic Acid Vaccines
mRNA vaccines have demonstrated safety and high efficacy in preventing SARS‑CoV‑2 infection, and their development was based on earlier RSV research. Both mRNA COVID‑19 vaccines express stabilized SARS‑CoV‑2 pre‑F spike proteins, a design that drew on the successful use of RSV pre‑F protein as a vaccine antigen. Extensive research on RSV vaccine‑associated enhanced respiratory disease also provided an important foundation for the rapid development of COVID‑19 vaccines and offered regulatory guidance on vaccine safety. After the successful scale‑up and establishment of supply chains for mRNA vaccines, mRNA technology has become a new platform for the development of other vaccines, including RSV vaccines.
Conclusion
Over the past decade, RSV vaccine development has shifted from empirical design to rational design, achieving remarkable progress. Multiple late‑stage clinical trial failures have laid the foundation for future success, particularly by supporting the pre‑F protein as a vaccine antigen, emphasizing multi‑season trials, and monitoring viral resistance to monoclonal antibodies, thereby advancing the development of both vaccines and monoclonal antibodies. Currently, RSV vaccine development has entered a critical phase, with nine vaccine candidates in Phase III trials. Monoclonal antibodies such as nirsevimab are expected to gain market access within the next one to two years. Meanwhile, maternal vaccines and vaccines for the elderly are also in late‑stage trials. These advances signal that RSV prevention is within reach. However, even as next‑generation monoclonal antibodies are poised for approval, palivizumab may continue to serve as a backup strategy until new antibodies are widely available globally.
In the future, global access to RSV vaccines and monoclonal antibodies still faces many challenges, especially in low‑ and middle‑income countries. Monoclonal antibodies may be an ideal option in these regions before vaccines become widely available, particularly where maternal immunization has not yet been implemented. To ensure the global success of RSV prevention, priority must be given to issues such as viral resistance monitoring, vaccine pricing, and implementation in high‑risk areas. Multiple immunization strategies, including infant immunoprophylaxis, maternal vaccines, and live‑attenuated vaccines for children, may together provide a comprehensive solution for RSV prevention, offering diverse application scenarios and preventive advantages. Canton Biologics has the capability to develop RSV vaccines and can contribute to the field of RSV prevention, further advancing vaccine accessibility.
References
-
Mazur NI, Terstappen J, Baral R, et al. Respiratory syncytial virus prevention within reach: the vaccine and monoclonal antibody landscape. Lancet Infect Dis. 2023;23(1):e2-e21. doi:10.1016/S1473-3099(22)00291-2
-
Herring WL, Zhang Y, Shinde V, Stoddard J, Talbird SE, Rosen B. Clinical and economic outcomes associated with respiratory syncytial virus vaccination in older adults in the United States. Vaccine 2021; 40: 483–93.
-
van der Fits L, Bolder R, Heemskerk-van der Meer M, et al. Adenovector 26 encoded pre-F conformation stabilized RSV-F protein induces long-lasting Th1-biased immunity in neonatal mice. NPJ vaccines 2020; 5: 49.
-
Tang A, Chen Z, Cox KS, et al. A potent broadly neutralizing human RSV antibody targets conserved site IV of the fusion glycoprotein. Nat Commun 2019; 10: 4153.
-
Karron RA, Atwell JE, McFarland EJ, et al. Live-attenuated Vaccines Prevent Respiratory Syncytial Virus-associated Illness in Young Children. Am J Respir Crit Care Med 2021; 203: 594–603.
-
Crank MC, Ruckwardt TJ, Chen M, et al. A proof of concept for structure-based vaccine design targeting RSV in humans. Science 2019; 365: 505–09.
-
Langley JM, MacDonald LD, Weir GM, et al. A respiratory syncytial virus vaccine based on the small hydrophobic protein ectodomain presented with a novel lipid-based formulation is highly immunogenic and safe in adults: a first-in-humans study. J Infect Dis 2018; 3: 378–87.
-
Marcandalli J, Fiala B, Ols S, et al. Induction of potent neutralizing antibody responses by a designed protein nanoparticle vaccine for respiratory syncytial virus. Cell 2019; 176: 1420–31.e17.

