Recombinant protein vaccines, also known as genetically engineered recombinant subunit vaccines, essentially express large quantities of pathogen antigenic proteins in engineered cells (e.g., CHO cells) using genetic engineering methods, followed by purification of the protein for vaccine construction. This type of vaccine offers advantages such as high safety, relatively mature technology, and low production cost. Compared to vaccines formulated from whole pathogens, recombinant protein vaccines focus the immune response on recognizing a small set of target antigens and are safer for immunocompromised individuals. However, the development of recombinant protein vaccines presents challenges including optimizing production systems, ensuring product stability and purity, and scaling up the manufacturing process. Addressing these challenges to successfully produce safe, effective, and high‑quality vaccines has become a major research focus.
Challenge 1: Optimizing the Expression System
Increasing the yield and quality of the target antigen is one of the keys to developing recombinant protein vaccines. To address this, Canton Biologics selects appropriate expression systems (e.g., bacteria, yeast, or mammalian cells) and optimizes cell culture conditions to ensure a robust system that stably produces large quantities of antigen.
Solutions
Cell Line Development (CLD)
-
Enhance cell line productivity through gene amplification, metabolic engineering, and high‑yield clone screening.
-
Use high‑performance host cell lines such as CHOzen® (CHO‑K1) combined with efficient vectors like Canvector® to achieve high expression and rapid screening.
-
Leverage the DTEasy platform to solve difficult‑to‑express protein problems, customize protein sequences, and increase titer and stability.
Upstream Process Solutions
-
High productivity: Achieve high yield and high quality of recombinant proteins and fusion proteins through media and process optimization.
-
Robust scale‑up platform: Ensure seamless transition from small‑scale to large‑scale production using advanced bioreactors and control strategies to maintain consistency and quality.
-
Process intensification: Implement high‑density cell culture, perfusion, and continuous processing to optimize nutrient supply, significantly increase productivity, shorten production time, and ensure flexibility and consistency.
Challenge 2: Ensuring Product Purity and Stability
The efficacy and safety of recombinant protein vaccines depend on their purity and stability. The purification process must remove impurities such as host cell proteins, host cell DNA, and endotoxins, while maintaining antigen stability during production, storage, and transportation.
Solutions
Downstream Purification Strategies
-
Employ multiple chromatography techniques (e.g., affinity, ion exchange, and size exclusion chromatography) combined with a high‑throughput resin screening platform to rapidly develop and optimize chromatography processes, achieving >95% purity and reducing the risk of adverse reactions in patients.
-
Effectively remove host cell proteins (HCP) and host cell DNA (HCD) through advanced purification processes to reduce immunogenicity and ensure safety.
-
Use optimized strategies to improve the stability of intermediates while ensuring effective virus inactivation and strictly controlling the risk of viral contamination.
Stability Strategies
-
Use stabilizers such as sugars and amino acids, optimize buffer conditions, and employ lyophilization technology to prevent protein degradation and aggregation during production and storage.
-
Use advanced analytical methods such as HPLC, mass spectrometry, and dynamic light scattering (DLS) to characterize purity, stability, and structural integrity, identifying and addressing potential stability issues.
Challenge 3: Scaling Up Production
Scaling up a process from a research laboratory to commercial scale presents many challenges. The process must be scalable and robust, capable of producing large volumes of vaccine to meet global demand while ensuring consistent quality and compliance with stringent regulatory standards.
Solutions
Manufacturing Science and Technology (MSAT)
MSAT is a core department at Canton Biologics for production scale‑up. The MSAT team ensures smooth transfers from laboratory to pilot scale to commercial production. Through detailed gap analysis, comprehensive risk assessment, and control of critical process parameters, they guarantee 100% successful process scale‑up and technology transfer.
Single‑Use Technology
Use single‑use bioreactors and systems to provide production flexibility, rapidly adjust output, reduce the risk of cross‑contamination between batches, and simplify cleaning processes. Single‑use technology reduces equipment preparation time and maintenance costs, improves production efficiency, ensures product quality consistency, and is widely accepted by regulatory agencies.
Quality by Design (QbD)
The QbD approach ensures that quality control is embedded in the production process from the very beginning. By understanding the relationship between process parameters and product quality attributes, critical process parameters are identified and controlled to ensure consistent production of high‑quality vaccines.
Regulatory Compliance and Documentation
Canton Biologics ensures that all scale‑up operations comply with GMP and other regulatory standards. Through detailed documentation and validation studies, process robustness and consistency are demonstrated, guaranteeing product compliance and manufacturing reliability.
Canton Biologics Recombinant Protein Vaccine Case Studies
Case Study 1
For a certain bivalent vaccine produced by Canton Biologics, both yield and quality were significantly higher than expected (expression level 5 g/L, SEC purity >99%).
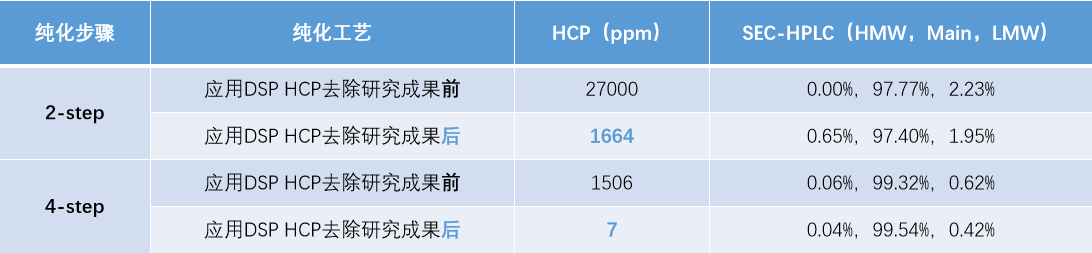
Whether using a 2‑step or 4‑step chromatography process, after applying DSP’s research results on HCP removal, the residual HCP in the product was greatly reduced while SEC purity remained unchanged. Process‑related impurities were effectively cleared (HCP below 10 ppm; a significant reduction compared to 900 ppm for a similar marketed product).
(Images placeholder – see original)
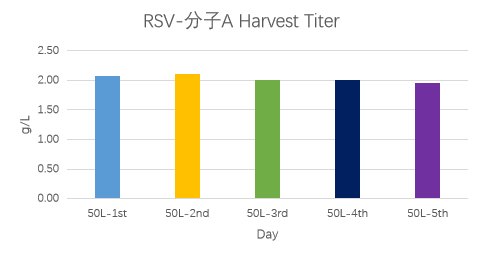
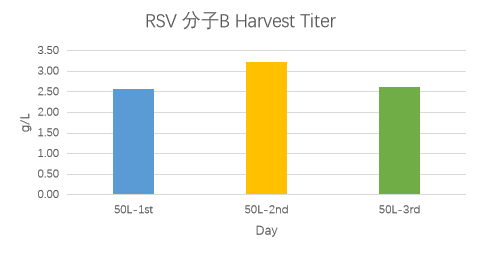
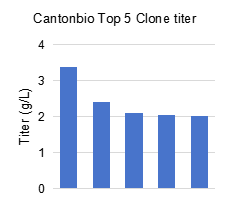
For RSV molecule A, expression levels across five batches were approximately 2 g/L; for RSV molecule B, the expression level reached up to approximately 3.2 g/L.
Case Study 2
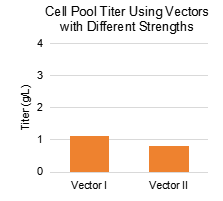
Canton Biologics’ Canvector® platform includes multiple recombinant protein vectors with different strengths. By running parallel tests to select the most suitable vector, expression levels can be effectively increased. Clone expression levels were 2‑3 times higher compared to similar vaccines (0.8‑1.0 g/L).
(Images placeholder – see original)
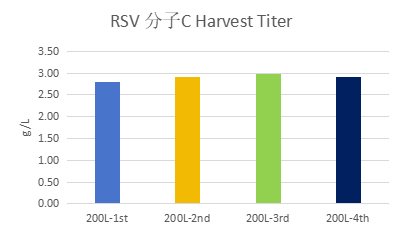
Expression levels for different RSV molecules were all above 2 g/L, reaching up to 3 g/L.
Conclusion
In summary, the CMC development of recombinant protein vaccine drugs is a complex and multi‑faceted process involving several stages of activities. It includes activities related to the creation, testing, and approval of the drug manufacturing process, ensuring that the vaccine meets safety, efficacy, and quality standards. Each stage presents unique challenges that must be effectively addressed to ensure successful vaccine development and application. Based on extensive experience in vaccine development, Canton Biologics has successfully navigated the complexities of various vaccine projects, including COVID‑19 vaccines, respiratory syncytial virus (RSV) vaccines, shingles vaccines, and others, consistently meeting regulatory standards and achieving high potency.

